Resources
The resources area of Bossklein.com contains a variety of useful technical and marketing materials to help provide more information regarding our infection control range of products.
This information has been designed to provide additional support to our worldwide network of dealers and distributors. By providing this additional support we hope we can help you effectively develop your business and keep the pressure growing around the world in the fight against infection with Bossklein.
Please use the information below to find out more and to download and/or view. Please be aware that some downloads are password protected so please ask your local Directa Dental Group sales representative or contact info@bossklein.com to unlock.
Don’t see what you need? Please don’t hesitate to contact us and we would be happy to discuss any individual needs or requests you may have.
Full Technical Brochure
See the full details of the Bossklein Complete Hygiene Solutions range with our new technical brochure.
Useful product information such as contact times, packing information, test standards, volumes/variations etc can be found inside to help both established and potential partners.
View using the interactive booklet on the right – drag pages from the corner to turn or use the arrows. Full screen mode is recomended.
Alternatively, click here to download the standalone PDF.
Image Libraries
Print ready product images
300dpi high resolution images. White (jpeg) or transparent (png) backgrounds. Ideal for all print and web related applications.
Click here to download (500mb+)
Web only product images
72dpi online friendly images. White backgrounds (jpeg) Ideal for all web applications.
Click here to download (30mb+)
Product Guides and Presentations
Bossklein – An introduction
A short introduction to the Bossklein Infection Control brand of products.
Click the image or here to view/download
Instrument Care
An overview of Bossklein Instrument Care infection control products.
Click the image or here to view/download
Surface Care
Take a look at the Bossklein Surface Care infection control product range.
Click the image or here to view/download
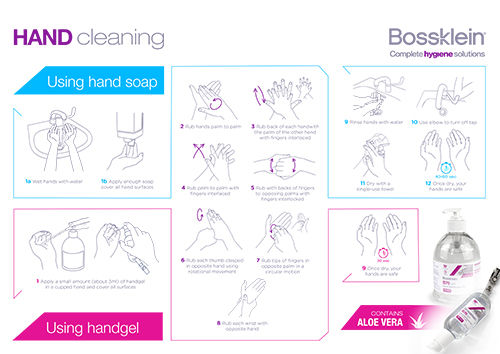
Special Areas + Skin & Hands
An overview of Bossklein Special Areas and Hand Care infection control products.
Click the image or here to view/download
Packaging and Accessories
A variety of different packaging options are available in the Bossklein range.
Click the image or here to view/download
Posters
Large format Bossklein posters for each area of infection control. Can be added to frames. Ideal for exhibitions, meeting rooms and wall mounting anywhere. Various sizes. 300dpi High resolution PDFs.
Click on an image to view/download.
Supporting Product Documentation
DAILYasp – A product focus
Find out more about this market leading aspirator cleaner and disinfectant with our Bossklein DAILYasp quick guide.
Click the image or here to view / download
IDactiv vs Dis Bur Ready
IDactiv was designed to replace the old Bossklein DisBur Ready solution, compare the two solutions in this interactive booklet.
Click the image or here to view / download
ECO-XL Eco Facts
Understand the benefits of switching to our eco-friendly biodegradable wipes with this mini booklet. Wipes made from natural plant based fibers
Click the image or here to view/download
Videos
DAILYasp – How it works
Manual cleaning with IDactiv
S Wipe technique V-WIPE ZERO
B70gel – Correct hand care
ULTRAbio – In the ultrasonic bath
Low streak finish – ECO-XL